有机化学方程式(选修五)
烷烃(甲烷为例)
1.CH4+Cl2 光照 CH3Cl+HCl ….
2.CnH2n+2+(3n+1)/2O2 点燃2+(n+1)H2O
烯烃(乙烯为例)
1.CH2=CH2+Br2→CH2Br—CH2Br
2.CH2=CH2+H2CH
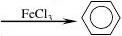
3CH2OH
3.CH2=CH2+HCl 3CH2Cl
4.CH2=CH2+HCH3—CH3 △ 催化剂 催化剂催化剂
5.nCH2=CH2催化剂 2—CH 6.CnH2n+3n/2O2点燃nCO2+nH2O
炔烃(乙炔为例)
1.CH≡CH+2H2CH3—CH3 △ 催化剂
2.CH≡CH+HCl 催化剂 CH2=CHCl
△
3.CH≡CH+Br2 → CHBr=CHBr
4.CH≡CH+2Br2→CHBr2—CHBr2
5.nCH2=CHCl 2—
6.C4H10C2H4+C2H6 加热、加压 催化剂 苯
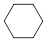
1. +HNO3 NO2+H2O 55~60℃ 浓硫酸
2. +Br2 (液溴Br + HBr
3. +3H2
△ 催化剂 1
苯的同系物(甲苯为例)
33 O2NNO2
NO2
COOH 1. +3HNO3 +3H2O 55~60℃ 浓硫酸 3 2、
KMnO4(H+)
卤代烃(溴乙烷为例)
1.CH3CH2Br+NaOH H2O CH3CH2OH+NaBr
△
醇
2.CH3CH2Br+NaOH △ 2=CH2↑+NaBr+H2O 醇
1.2CH3CH2OH+2Na → 2CH3CH2ONa+H2↑
2.CH3CH2OH+3CH2Br+H2O
浓硫酸 △
3.CH3CH2OH 170℃ CH2=CH2↑+H2O
4.2CH3CH2OH 3CH2—O—CH2CH3+H2O 140℃ 浓硫酸
5.2CH3CH2OH+O22CH3CHO+2H2O △ 催化剂
6.2CH3CH(OH)CH3+O23COCH3+H2O △
7. C(CH3)3 OH+O2 催化剂 很难被氧化
△ 催化剂
酚(苯酚为例)
1. +2Na→2 + H2↑
2. + NaOH → +H2O
2
3. +CO2 + H2O → + NaHCO3
4. +Na2CO3→ + NaHCO3
Br Br—5. +3Br2 → ↓ +3HBr
Br
6. 溶液呈紫色
醛(乙醛为例)
1.CH3CHO+H2 CH3CH2OH △ 催化剂
2.2CH3CHO+O2 2CH3COOH △ 催化剂
3.CH3CHO+2[Ag(NH3)2]OH 3COONH4+3NH3+H2O+2Ag↓
4.CH3CHO+2Cu(OH)2+NaOH CH3COONa+Cu2O↓+3H2O 羧酸 酯
1.CH3CHOOH+CH3CH2OH
2.CH3COOCH2CH3+H2O
3. CH3COOCH2CH3+NaOH浓硫酸 △ 稀硫酸 △ △ CH3COOCH2CH3+H2O CH3CHOOH+CH3CH2OH CH3CHOONa+CH3CH2OH 3
第二篇:高中化学选修5化学方程式总汇
有机化学方程式(选修五)
烷烃(甲烷为例)
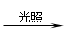 1.CH4+Cl2 CH3Cl+HCl ….
1.CH4+Cl2 CH3Cl+HCl ….
 2.CnH2n+2+(3n+1)/2O2 nCO2+(n+1)H2O
2.CnH2n+2+(3n+1)/2O2 nCO2+(n+1)H2O
烯烃(乙烯为例)
1.CH2=CH2+Br2→CH2Br—CH2Br

 2.CH2=CH2+H2O CH3CH2OH
2.CH2=CH2+H2O CH3CH2OH
3.CH2=CH2+HCl CH3CH2Cl
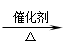 4.CH2=CH2+H2 CH3—CH3
4.CH2=CH2+H2 CH3—CH3


 5.nCH2=CH2 [CH2—CH2]n
5.nCH2=CH2 [CH2—CH2]n
 6.CnH2n+3n/2O2 nCO2+nH2O
6.CnH2n+3n/2O2 nCO2+nH2O
炔烃(乙炔为例)
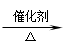 1.CH≡CH+2H2 CH3—CH3
1.CH≡CH+2H2 CH3—CH3
 2.CH≡CH+HCl CH2=CHCl
2.CH≡CH+HCl CH2=CHCl
3.CH≡CH+Br2 → CHBr=CHBr
4.CH≡CH+2Br2→CHBr2—CHBr2
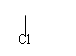


 5.nCH2=CHCl [CH2— CH ]n
5.nCH2=CHCl [CH2— CH ]n
6.C4H10 C2H4+C2H6
苯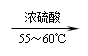

 1. +HNO3 —NO2+H2O
1. +HNO3 —NO2+H2O


 2. +Br2 (液溴) —Br + HBr
2. +Br2 (液溴) —Br + HBr

 3. +3H2
3. +3H2
 苯的同系物(甲苯为例)
苯的同系物(甲苯为例)

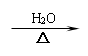
 1. +3HNO3 +3H2O
1. +3HNO3 +3H2O
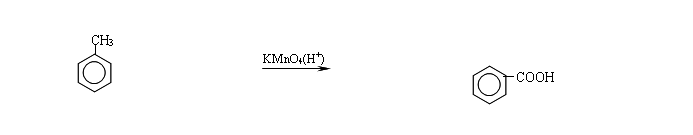
2、
 卤代烃(溴乙烷为例)
卤代烃(溴乙烷为例)
1.CH3CH2Br+NaOH CH3CH2OH+NaBr

2.CH3CH2Br+NaOH CH2=CH2↑+NaBr+H2O
醇
1.2CH3CH2OH+2Na → 2CH3CH2ONa+H2↑

 2.CH3CH2OH+HBr CH3CH2Br+H2O
2.CH3CH2OH+HBr CH3CH2Br+H2O
3.CH3CH2OH CH2=CH2↑+H2O
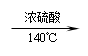 4.2CH3CH2OH CH3CH2—O—CH2CH3+H2O
4.2CH3CH2OH CH3CH2—O—CH2CH3+H2O
 5.2CH3CH2OH+O2 2CH3CHO+2H2O
5.2CH3CH2OH+O2 2CH3CHO+2H2O
 6.2CH3CH(OH)CH3+O2 2CH3COCH3+H2O
6.2CH3CH(OH)CH3+O2 2CH3COCH3+H2O
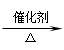 7. C(CH3)3 OH+O2 很难被氧化
7. C(CH3)3 OH+O2 很难被氧化

 酚(苯酚为例)
酚(苯酚为例)
1.2 +2Na→2 + H2↑


2. + NaOH → +H2O

 3. +CO2 + H2O → + NaHCO3
3. +CO2 + H2O → + NaHCO3

 4. +Na2CO3→ + NaHCO3
4. +Na2CO3→ + NaHCO3

 5. +3Br2 → ↓ +3HBr
5. +3Br2 → ↓ +3HBr
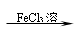
 6. 溶液呈紫色
6. 溶液呈紫色
醛(乙醛为例)
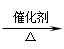 1.CH3CHO+H2 CH3CH2OH
1.CH3CHO+H2 CH3CH2OH
 2.2CH3CHO+O2 2CH3COOH
2.2CH3CHO+O2 2CH3COOH
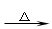 3.CH3CHO+2[Ag(NH3)2]OH CH3COONH4+3NH3+H2O+2Ag↓
3.CH3CHO+2[Ag(NH3)2]OH CH3COONH4+3NH3+H2O+2Ag↓
 4.CH3CHO+2Cu(OH)2+NaOH CH3COONa+Cu2O↓+3H2O
4.CH3CHO+2Cu(OH)2+NaOH CH3COONa+Cu2O↓+3H2O
羧酸 酯
 1.CH
3CHOOH+CH
3CH
2OH
1.CH
3CHOOH+CH
3CH
2OH
 CH
3COOCH
2CH
3+H
2O
CH
3COOCH
2CH
3+H
2O
 2.CH
3COOCH
2CH
3+H
2O
2.CH
3COOCH
2CH
3+H
2O
 CH
3CHOOH+CH
3CH
2OH
CH
3CHOOH+CH
3CH
2OH
 3. CH
3COOCH
2CH
3+NaOH
3. CH
3COOCH
2CH
3+NaOH
 CH
3CHOONa+CH
3CH
2OH
CH
3CHOONa+CH
3CH
2OH
