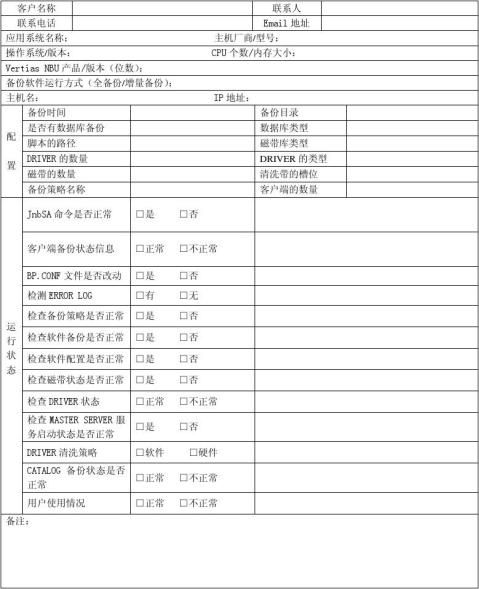
VERTIAS Net backup巡检报告
1/1
第二篇:Clinical Evaluation Report (Aspirator Type-XXX)-20xx 抽痰机临床评估报告-20xx
Sponsor
Name: ABC CO., LTD.
Address: XXXXXXXX
Product Information
Product Name: Aspirator
Product Type: (TYPE:XXX)
Abstract
This clinical evaluation report summarized the performance information for ABC CO., LTD. in order to demonstrate the clinical safety and effectiveness. We reviewed from 3 directions, the compliance to harmonized standard, equivalence comparison to marketed product, and literature. It is concluded that ABC CO., LTD. Aspirator (TYPE:XXX) is able to perform the intended use and does not compromise patient or other user's safety.
Intended Use
(TYPE:XXX) series suction machine the power suction pump with a suction cup and suction tube by suffering's Airway or respiratory support system in removing infected material. This suction machine is mainly to assist family members or care, alternate. Patients do accept tracheotomy suctioning action.
Biological Aspect
ABC CO., LTD. Aspirator (TYPE:XXX) do not need to the ISO10993 testing.
Harmonized standard
ABC CO., LTD. Aspirator (TYPE:XXX) had been tested according to EN60601-1 / EN60601-1-2, / ISO 10079-1 and was confirmed compliance by ABC CO., LTD. ‘s QA. There is no other specific international standard regarding these devices.
Equivalence comparison to marketed device
ABC CO., LTD. Aspirator (TYPE:XXX) had been compared to other two different brand products and the results were listed below:
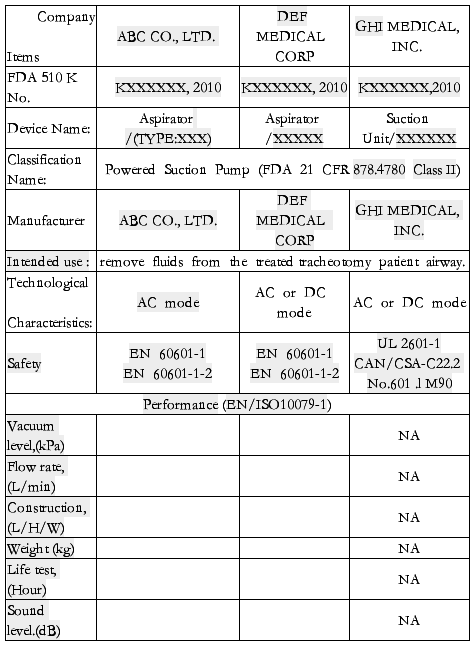
ABC CO., LTD. Aspirator (TYPE:XXX) is claimed to be substantial equivalent to other marketed products, with the same intended use and similar product design.
Literature review
We review recently 10 year’s literatures about aspirator (See References).
Post-Market Surveillance (PMS)
Same type(Apex) like ABC CO., LTD. Aspirator/(TYPE:XXX) has been introduced into the market for more than 12 years and distributed to more than 60 countries. Almost 500 thousand had been sold and no customer complaint regarding functions or safety has been reported to ABC CO., LTD.
Conclusion
It was concluded that ABC CO., LTD. Aspirator/(TYPE:XXX) is substantially equivalent to 510(k) cleared products, functions as intended, and without significant safety concerns. The assessment of user benefit to health from use as intended is much higher than the probable risks of injury or illness.
References
1. Creinin MD, Schwartz JL, Guido RS, Pymar HC. Early pregnancy failure—current management concepts. Obstet Gynecol Surv. 2001;56(2):105-113.
2. Blumenthal PD, Remsburg RE. A time and cost analysis of the management of incomplete abortion with manual vacuum aspiration. Int J Gynecol Obstet. 1994;45:261-267.
3. Fawcus S, McIntyre J, Jewkes RK, Rees H, Katzenellenbogen JM, Shabodien R, et al. Management of incomplete abortions at South African public hospitals. SAMJ. 1997;87(4):438-42.
4. Hemlin J, Moller B. Manual vacuum aspiration, a safe and effective alternative in early pregnancy termination. Acta Obstet Gynecol Scand. 2001;80:563-67.
5. Mahomed K, Healy J, Tandon S. A comparison of manual vacuum aspiration (MVA) and sharp curettage in the management of incomplete abortion. Int J Gynecol Obstet . 1994;46:27-32.
6. Westfall JM, Sophocles A, Burggraf H, Goldberg A. Manual vacuum aspiration for first-trimester abortion. Arch Fam Med. 1998;7:559-61.
Approved by: Prepared by:
