Date: 20##-05-22
Experimental title: Determination of reducing sugar content—DNS method
Principle: Determination of reducing sugar is the basic method for the determination of sugar content. Reducing sugar is free aldehyde or ketone containing sugar. Monosaccharides are reducing sugars, disaccharides and polysaccharides are not necessarily reducing sugar. Lactose and maltose are reducing sugars, sucrose and starch non-reducing sugar. The solubility of the different use of sugar, you can sample monosaccharide, disaccharides, and polysaccharides were extracted. (Not reducing) disaccharides and polysaccharides can be double-acid hydrolysis method. Outing to double sugar and reducing the degradation into a single sugar determination. In the sample were calculated reducing sugars content. Content of reducing sugar with glucose instead.
Procedures:
& Reagent preparation:
Ø DNS Reagent configuration
1) In NaOH solution, by adding dinitrosalicylic acid solution and potassium sodium titrate
2) NaOH solution by adding crystalline phenol, add water to dissolve, and finally set the volume.
3) Take (2) 69mL anhydrous sodium sulfite 6.9 g, to dissolve
4) To (3) into (1), the completely dissolved, stored in brown reagent bottle. At room temperature, place 7-10 days later to use
Ø Glucose standard solution configuration
Accurately weigh 100 mg of analytical grade glucose (pre-dried at 105 ℃ to constant weight), dissolved in distilled water with a small amount of volume to 100mL, refrigerator use.
& Operation methods
1) Take six clean test tubes, adding various reagents in the following order
i. Glucose solution
ii. Distilled water
iii. DNS reagent
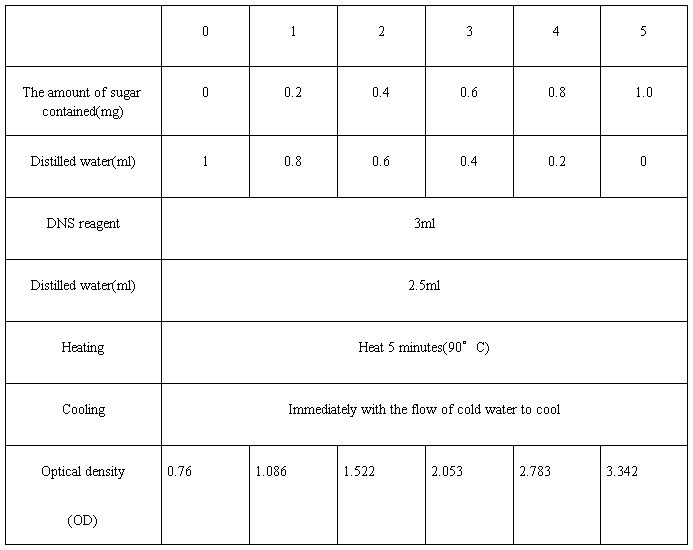
Results:
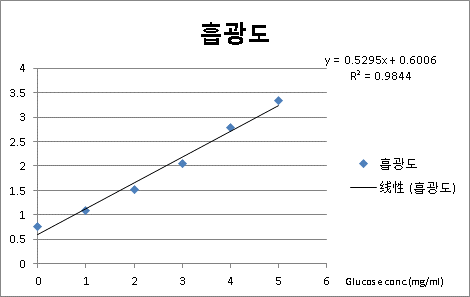
Mix the above solution, each tube after the photoelectric colorimeter (510 nm) for colorimetric determination of the blank control solution with zero adjustment, recording the optical density value. Glucose concentration as the abscissa, the ordinate is optical density standard curve plotted.
Discussion:
In alkaline solution, the reducing sugar into ene glycol (1,2 - ene glycol).
Ene glycol of various oxidants such as iron cyanide easily, 3,5 - dinitrosalicylic acid and Cu2 + oxidized to sugar acids. Cyanide and dynatron-salicylic acid salt reduction is the basis for quantitative determination of reducing sugar. Dinitrosalicylic acid reducing sugar and alkaline reagent is heated together to produce a brown-red amino compounds, at a certain concentration range, the depth of the color brown material degree proportional to the amount of reducing sugar. Therefore, we can determine the sample amount of reducing sugar and total sugar.
When you configure DNS reagent, to note 3,5 - dinitrosalicylic acid and NaOH, adding time must be very close, or to join the NaOH. Or will cause the precipitation of insoluble, leading to the preparation of the solution to fail. And the configuration process, the solution heating temperature should not exceed 50 degrees Celsius.
When you configure DNS to add potassium sodium titrate solution was to remove the inside of the dissolved oxygen, dissolved oxygen and more so, shorten the shelf life of a sample at different times, the results of the determination of large differences (sugar dissolved oxygen serious interference color). Anhydrous sodium sulfite is stable color, and phenol with the color of the color is more stable
Conclusion:
In the 3,5 - dinitrosalicylic acid reducing sugar in colorimeters, NDS is the main reagent. It is red and brown sugar reaction material. But the color just the words with the DNS, not a linear relationship between color and concentration. Or linear poor. Add the aim of increasing DNS phenol color reagent for quantitative determination of the linear relationship between reducing sugar concentration. The presence of phenol is essential. NDS does not participate in the color of phenol not quantitative. However, due to the air easily oxidized phenol case, the remaining phenol in order to ensure that all steps are not oxidized. Adding a sufficient amount of potassium sodium tartrate is to eliminate the impact of dissolved oxygen, sodium sulfite can be added to further enhance the stability of reagents.
DNS standard curve preparation, set a concentration gradient when the content should be zero DNS reagent as reference instead of using DNS as a reference.
Standard curve should be within the concentration range in a linear relationship. Best R = 0.999 or more. This of course does not rule out the possibility of a large concentration range. DNS and the role of the linear relationship between reducing sugar and the amount of reagent have nothing to do. 3,5 - dinitrosalicylic acid solution and reducing sugars (all monosaccharide and maltose) solution were reduced to brown red hot after the amino compounds, in a certain range, the amount of reducing sugar and brown colors of red objects Extent proportional to the depth of relationship.
第二篇:食品感官分析实验报告



二〇一三年四月
食品感官分析实验报告
排序试验
一、实验原理与目的
排序是使用标度的检验中最简单的一种方法,排序检验使用的数字类型是顺序数字,没有等距和成比例的意义,只排出样品的次序,不评价样品间差异的大小。该实验的目的是让评价员了解排序检验法的基本方法和适用范围。学会采用排序检验法对系列样品的某一感官品质的强弱进行比较。同时,学会对实验结果进行统计分析。 二、实验材料
(1)含糖量为2%、3%、4%、5%、6%的豆浆,纯净水 (2)检验容器:足量一次性水杯 三、实验步骤 1.被检样品的制备
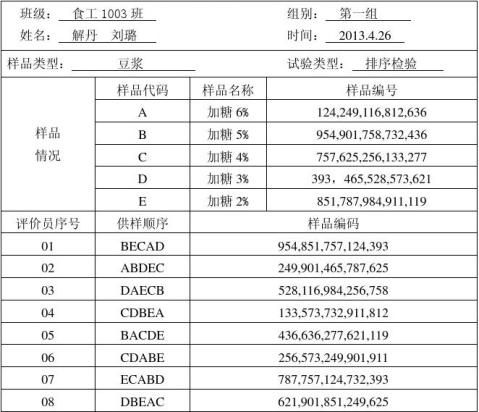
查随机数表,先获取所需的三位随机数,每个样品每位评价员准备一个编号,填入样品准备工作表。提供ABCDE五种豆浆,间五种样品按照试验设计进行随机组合,从实验室样品中制备相应数量的样品组,并在容器上对应编好号。
1
食品感官分析实验报告
2.品评检验
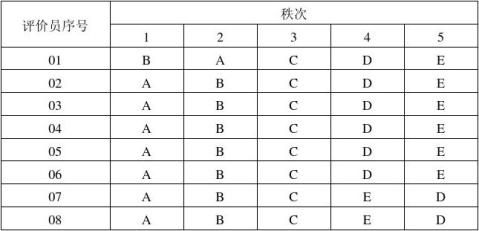
将按照准备表组合并标记好的样品连同问答表一起呈送给评价员。每个评价员每次得到随机提供的一组样品,依次品评后依检验的特性排列成一定的顺序,并填好问答表。
表2:排序检验法问答表
四、实验结果与分析
表3:评价员的排序结果

2
食品感官分析实验报告
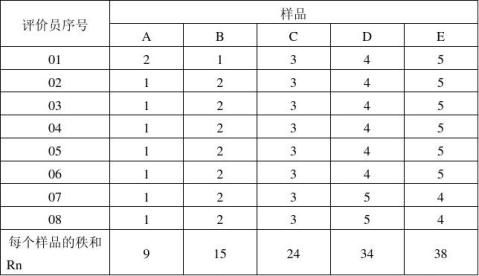
表4:样品的秩次与秩和
1.Friedman检验
F=12/8*5*(5+1)*(9?+15?+24?+34?+38?)—3*8*(5+1)=30.1
查表得,F=30.1大于P=5,J=8,α=0.01的临界值13.28,则判定样品之间有显著差异。 2.Kramer检验
依据评价员数为8、样品数为5得出显著水平为1%下的临界值为:
表5
的显著水平五个样品间有显著差异。
下段中,由于RA=9小于最小值Ri=15,RB=15等于最小值Ri=15,RD=34、RE=38大于最大值Ri=38,所以ABCDE五种样品可以划分为三个组: A B C D E 结论是:在1%的显著水平上,A、B样品最甜,C样品次之,D、E样品最不甜,且C样品在甜度上无显著性差异。
五、实验心得与体会
贾玉:在做排序实验时,感觉有的浓度差很大,有的还蛮小的,不过感觉都还蛮好分辨的,实验很快就做完了,豆浆也很快喝完了。
王慧玲:排序实验最甜的和最不甜的可以明显的感觉到,但是中间的甜度区分的不是很
3
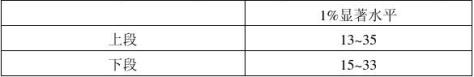
上段中,由于RA=9、RB=15小于最小Ri=13,RE=38大于最大Ri=35,所以在1%
食品感官分析实验报告
清楚,实验中可以感觉到每次评价完漱口显得十分的重要。
郎玉婷:通过这次试验,我了解了排序检验使用的数字类型是顺序数字,没有等距和成比例的意义,只排出样品的次序,不评价样品间差异的大小。同时学会了采用排序检验法对系列样品的某一感官品质的强弱进行比较。豆浆的中间两个样品比较难以分辨,其他的都比较简单。
李楠楠:本次实验的五杯豆浆中前三杯相对来说难区分一些,甜度都比较大,后三杯味道很淡容易区分一些,可以明显感觉到后三杯的香精味很浓,不过还是很容易区分的。 王冲:豆浆的甜度还是比较好分的,但是喝完甜的之后必须漱口,不然会串味,会尝不出来,漱口就比较好尝出来,不过太甜的两个不太好区分,太腻了
沈雅洁:实验还算比较好分辨,豆浆甜度最浓的两个有些不明显。其他的排序都还算容易的。豆浆有点腥口感不是很好。
辜文琦:实验时,对于豆浆的甜度排序,很甜和不甜的容易区分,但是对于甜度的排序,还是不是特别不敏感,甜度的排序有一两个不好区别开来,每次的漱口感觉有不少帮助。看来漱口对于判断还是很重要的。
张凤凤:通过第一次感官实验,对感官分析有了一定的了解和认识,这次做实验找到了些感觉。因为对甜比较敏感所以判断比较容易。
刘璐:实验准备和实验过程都进行得很顺利。实验准备过程有些麻烦耗时,需要打豆浆煮豆浆编号,并根据编号来添加各甜度的豆浆。通过实验我了解并掌握了排序检验的方法,对系列样品的某一感官品质的强弱进行比较,只排出样品的次序,不评价样品间差异的大小。评价员们能较准确地对豆浆甜度进行比较排序,但甜度相近的有些难分辨。实验结果的统计分析好难F值难算。
解丹:第一次做感官分析实验的组织者,感觉还是挺有意义的。在组织过程中,准备样品的过程有些复杂,排号编码是最麻烦的,不过还好在我和另一位组织者的合作下顺利完成了样品的制备。在组织评价员品评的过程中评价员表现都很好,都有按照试验要求做实验,最后我们统计出的结果也是可观的。我觉得做实验组织者可以帮助我们把实验和理论知识学习的更透彻,更明白,当然在以后的实验中也要认真学习,不能马虎。 4
