xxxxxxxxxx Hospital
中国石油 Chest X-Ray Report
Date 2013/08/16
Gender: Female Age:23 DOB:1990/10/22 No :2013081618 Name:
Projection Position: Chest Film in upright PA view Imaging findings:
1、Thorax is symmetrical in bilateral.
2、Trachea is in the middle.
3、Both lung field are clear.
Hilus of the lung are not enlargement.
4、The size and shape of heart are normal.
Cardiothoracic ratio is 0.5.
5、Mediastinum is normal.
6、Both diaphragmas are normal.
Diagnosis conclusion :
Examination of chest X-ray is normal.
Examiner:
Reviewer: Date:
第二篇:英文科学报告
Science investigation
----Hai.Quan

Rusting is the corrosion of iron and readily occurs in the alloy steel. The formation of reddish brown flakes which loosely adheres to the iron is called rust. This investigation is investigating about velocity of the rust. The title been investigated: Iron can react with water and oxygen to produce the rust, and the task in this investigation is to find out the production of rust in different solutions on iron. The purpose of this investigation: Iron rust makes buildings, boats, planes and any other things made by metal fragile. So prevent rusting is a very important thing for people to do. In this investigation, we are going to investigate what makes more rust, and what can prevent rusting. And the result of this investigation will benefit people in the daily life. Hypothesis: People predict that salt will increase the rusting speed, and oil helps preventing rusting. Equipment used:
Three one-off paper cups, dozens of nails, measuring cup (measure water capacity), camera (taking photos), and computer (settle the statistics, combine and evaluate the situations in the investigation) The reaction in the iron rusting: Iron can be rust, and it can rust with oxygen and water together when it is in the usual circumstance.Chemical formula of rust or dehydrated iron (III) hydroxide is Fe2O3.nH2O which is formed through a series of chemical reactions.
Risk assessment: ? It will be a bit dangerous when people penetrate the nails through the paper in order to make them stand. It is dangerous because nails might pierce the skin when people do it. The using of rubber glove is a good way to protect hands no harm. ? The iron rust might erode people’s hand, so when people touch the iron rust (especially dissolved in the water), they should wear rubber gloves. The way to prevent the variables: 1 Three cups must be putted in the exactly same place; otherwise, the different evaporation of water might influence the result. 2 Three cups must have the exactly same capacity of water. The way to measure the same capacity of water is to use the measuring cup. 3 Different numbers of nails might produce different quality of rust. In order to make the quality of rust fair, people must put exact same number of nails into the cup. The process of this investigation:
1.
2.
3.
4. Three same shapes, one-off paper cups with the same cubage. The first cup has 50ml water. The second cup has 50ml water plus 15%of salt. The third cup has the same water and nails cover oil. Fold three pieces of tissues, and penetrate the same number of nails in each in order to make nails stand. Put three pieces of tissues with nails into three cups of water slowly. Keep these cups in the same circumstance for ten days to observe the result.
Investigation diary
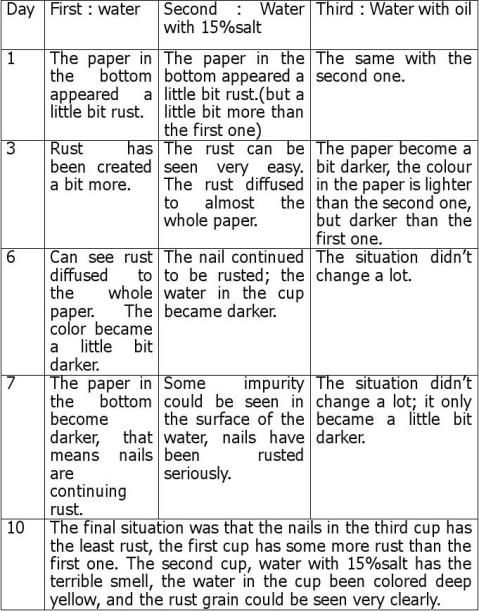

The graph of iron rusting
Photos taken during the investigation:



Discussion of this investigation: ? The design of the experiment is successful. First, the designer cleared the purpose of the investigation, and he listed tools which need in the investigation. Second, the designer wrote the steps of this research completely, it makes people easy to follow. ? The suggestion of modifications is to set some more cups. More cups can reduce the probability of special phenomena happens. ? The explanation of results is good. The investigation maker used photo, table and graph describes the result of the experiment. In the table of the result, the investigation maker noted the rust grade each day, and describes the colour and character of the rust in each cup, details are also noted. So it is also successful. ? For further investigation, I suggest that some more photos are necessary. Words cannot describe anything; photo recording may help recording more information for people to use.
Conclusion of the investigation: 1 Can oil stop rusting in iron? As the ‘iron rusting graph’ shown, nails in the third cup (oil) rusted a bit faster than the first one (only water), but after approximately five days, the third cup’s rusting speed decreased, and after ten days, the third cup (oil) had the least rust. So that shows oil can stop rusting. 2 Will salt increase the velocity of iron rusting? The answer of this question is ‘yes’. In the rusting diagram, the line of the second cup (15%salt) is always higher than another two lines. That means the nails in the salt water rusted faster than another two cups of nails. This proved that salt will increase the velocity of iron rusting. 3 Why does the third cup have more rust than the first one in the first four days? By using the internet, I found that oil can stop the iron rusting. The conditions of iron rusting are oxygen and water. Iron would rust only water can oxygen come together. Oil can stop the oxygen mixing with water, so theoretically, oil can stop rusting. But oil has colour, generally light yellow. I used nut’s oil from the kitchen, so the colour is a little bit deep. With some reasons with the light, the nails will show more rust. So I think that is the reason why I saw there was more rust on the third cup. 4 Does the hypothesis accept referring to results? The hypothesis is the same as the investigation result. As the questions answering shown above, oil stops rusting, and salt increases the velocity of rusting. The hypothesis is right and successful.
Bibliography reference related to this investigation: ? <Interview, David Des Marais> ? <inorganic chemistry> ? <Ullmann’s encyclopedia of industrial chemistry> ? <corrosion>
