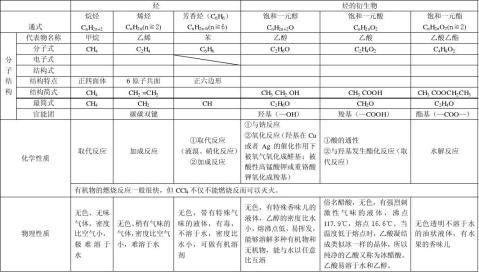
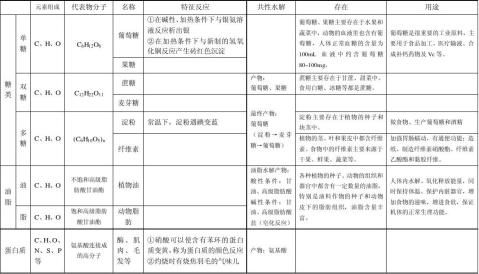
无机方程式(必修二)
4Li+O2 2Li2O
2Li2O
2Na+O2 Na2O2
Na2O2
2Na+2H2O=2NaOH+H2↑
2K+2H2O=2KOH+H2↑
2NaBr+Cl2=2NaCl+Br2
2NaI+Cl2=I2+2NaCl
Br2+2KI=I2+2KBr
2Na+Cl2 2NaCl
2NaCl
原电池的电源反应:(铜锌原电池,电解液稀硫酸)
锌片:Zn - 2e- = Zn2+(氧化反应)
铜片:2H+ + 2e- = H2↑(还原反应)
钢铁吸氧腐蚀的正负极反应
负极:Fe-2e-=Fe2+;正极:2H2O+4e-+O2=4OH-
氢氧燃料电池在氢氧化钾溶液中的正负极反应
负极;2H2-4e-+4OH-=4H2O;正极:O2+4e-+2H2O=4OH-
CH4(g) + 2O2(g)  CO2(g) + 2H2O(l)
CO2(g) + 2H2O(l)
CH4+Cl2 CH3Cl+HCl
CH3Cl+HCl
C2H4+3O2 2CO2+2H2O
2CO2+2H2O
CH2=CH2 + Br-Br→ CH2Br-CH2Br
2C6H6+15O2 12CO2+6H2O
12CO2+6H2O

 +HO-NO2
+HO-NO2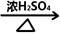
 +H2O+3H2
+H2O+3H2
2CH3CH2OH+2Na
 2CH3CH2ONa+H2↑
2CH3CH2ONa+H2↑
CH3CH2OH+3O2 2CO2+3H2O
2CO2+3H2O
2CH3CH2OH+O2 2CH3CHO+2H2O
2CH3CHO+2H2O
CH3CO-OH+H-O-C2H5 CH3COOC2H5+H2O
CH3COOC2H5+H2O
C12H22O11+H2O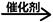 C6H12O6(葡萄糖)+C6H12O6(果糖)
C6H12O6(葡萄糖)+C6H12O6(果糖)
淀粉或纤维素的水解反应:(C6H10O5)n+nH2O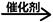 nC6H12O6
nC6H12O6
2HgO 2Hg+O2↑
2Hg+O2↑
2Ag2O 4Ag+O2↑
4Ag+O2↑
MgCl2(熔融) Mg+Cl2↑
Mg+Cl2↑
2Al2O3(熔融) 4Al+3O2↑
4Al+3O2↑
2NaCl(熔融) 2Na+Cl2↑
2Na+Cl2↑
铝热反应:Fe2O3+2Al 2Fe+Al2O3
2Fe+Al2O3
水煤气的制法:C(s)+ H2O(g)  CO(g) + H2(g)
CO(g) + H2(g)
C16H34 C8H18+C8H16
C8H18+C8H16
C8H18 C4H10+C4H8
C4H10+C4H8
C4H10 CH4+C3H6
CH4+C3H6
C4H10 C2H4+C2H6
C2H4+C2H6
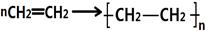
第二篇:高中化学必修2第三章有机化合物