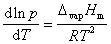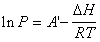实验二 液体饱和蒸气压的测定
Measurementof Saturated Vapor Pressure
王暮寒 PB10207067
中国科学技术大学生命科学学院
Muhan Wang PB10207067
School of Life Science, University of Science & Technology of China, Hefei
Email:wangmh@mail.ustc.edu.cn
2012.5
【关键词】
饱和蒸汽压
克拉伯龙-克劳修斯方程
温度
摩尔汽化热
【KEYWORDS】
Saturated vapor pressure
Claperon-Clasius Equation
temperature
Molar Heat of vaporization
【摘要】
根据克拉贝龙-克劳修斯方程,在一定温度范围内,对于纯液体的炮和蒸汽压P与温度T,lnP与1/T之间存在线性关系。利用动态法测定不同温度T下环己烷的饱和蒸汽压p,得出克拉贝龙-克劳修斯方程,从而求算环己烷的摩尔气化热,求得环己烷在大气压下的正常沸点。
【ABSTRACT】
According to the Claperon-Clasius Equation, there is a linear relation between T, the temperature of a fluid and p, the saturated vapor pressureof it.By measuring the saturated vapor pressure of cyclohexaneunder different temperatures, we could verify the linear relation, and further more, calculate the molar heat of vaporization along with the boiling point of cyclohexane.
…… …… 余下全文




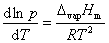 (1)
(1)  (2)
(2)  宁 波 工 程 学 院
宁 波 工 程 学 院